MYR301 STUDY INFORMATION
STUDY DESIGN
The MYR301 study was a multicenter, open-label, randomized, Phase 3 study in adults
with chronic HDV with or without cirrhosis (n=100)1
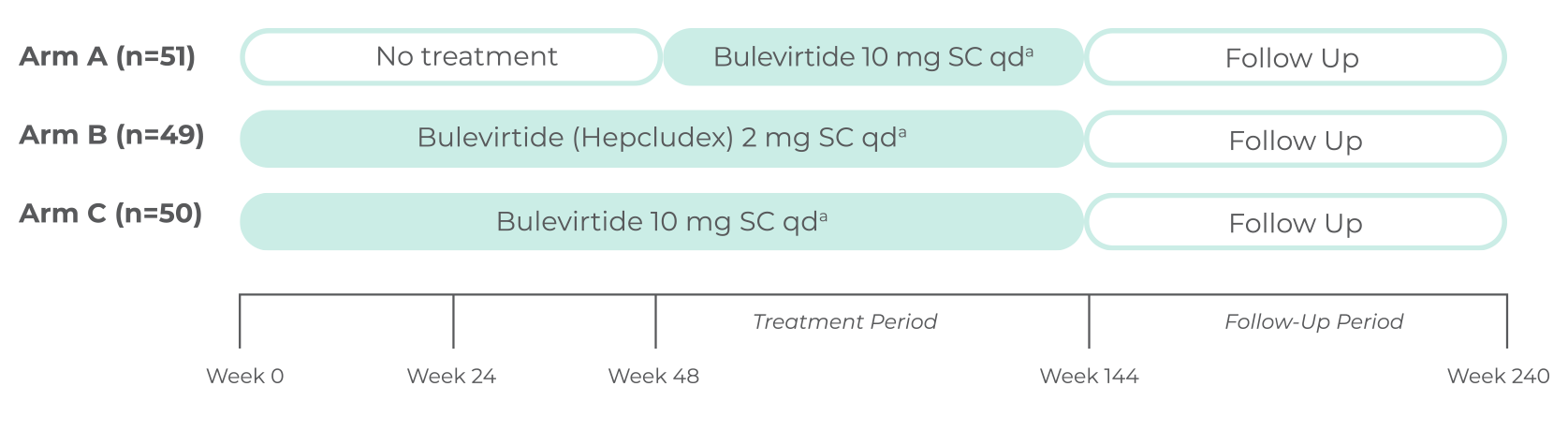
a2-mg Hepcludex is the approved dosage. Other doses have been studied as part of the dose-finding study.
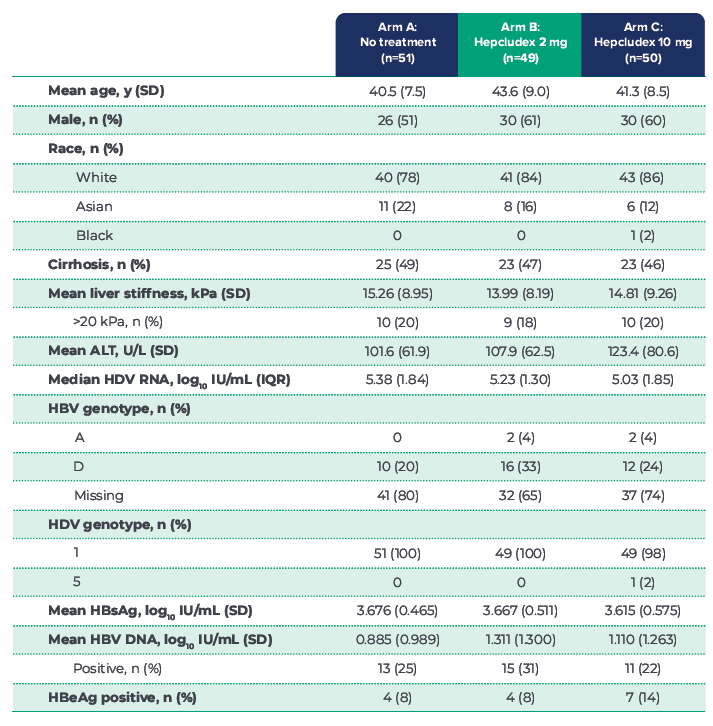
BASELINE CHARACTERISTICS

PRIMARY ENDPOINT
A combined response of undetectable
HDV RNA or decrease of ≥2 log10 IU/mL
from baseline and ALT normalization at
48 weeks
SELECT SECONDARY ENDPOINTS
- Undetectable HDV RNA at Week 48
- ALT normalization at Week 48
- Sustained virologic responses at Weeks 24 and 48
PRIMARY ENDPOINT
A combined response of undetectable
HDV RNA or decrease of ≥2 log10 IU/mL
from baseline and ALT normalization at
48 weeks
SELECT SECONDARY ENDPOINTS
- Undetectable HDV RNA at Week 48
- ALT normalization atWeek 48
- Sustained virologic responses at Weeks 24 and 48
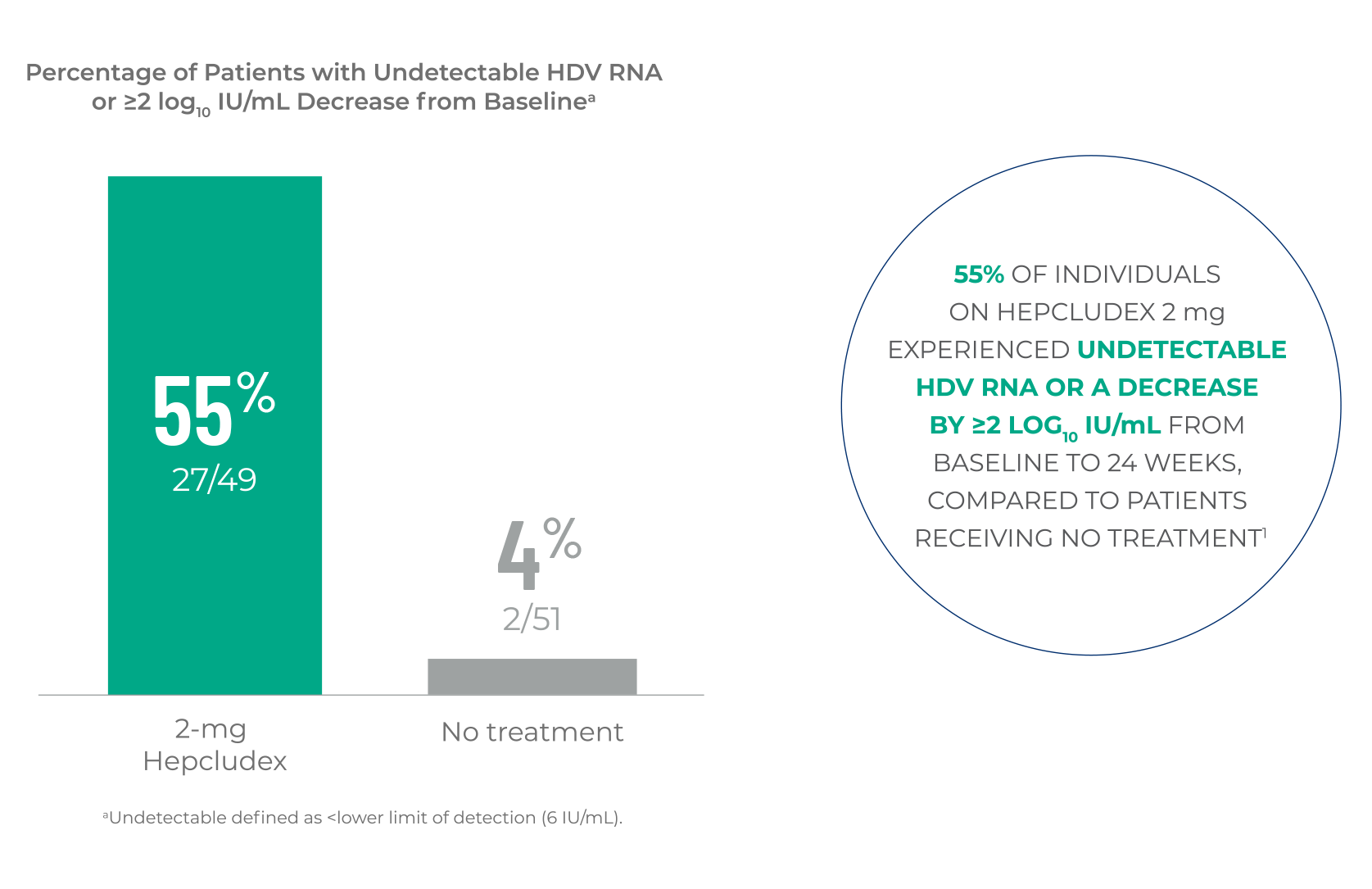
HDV RNA REDUCTION
Hepcludex demonstrated a reduction in HDV RNA levels at Week 24
compared to no treatment1,a
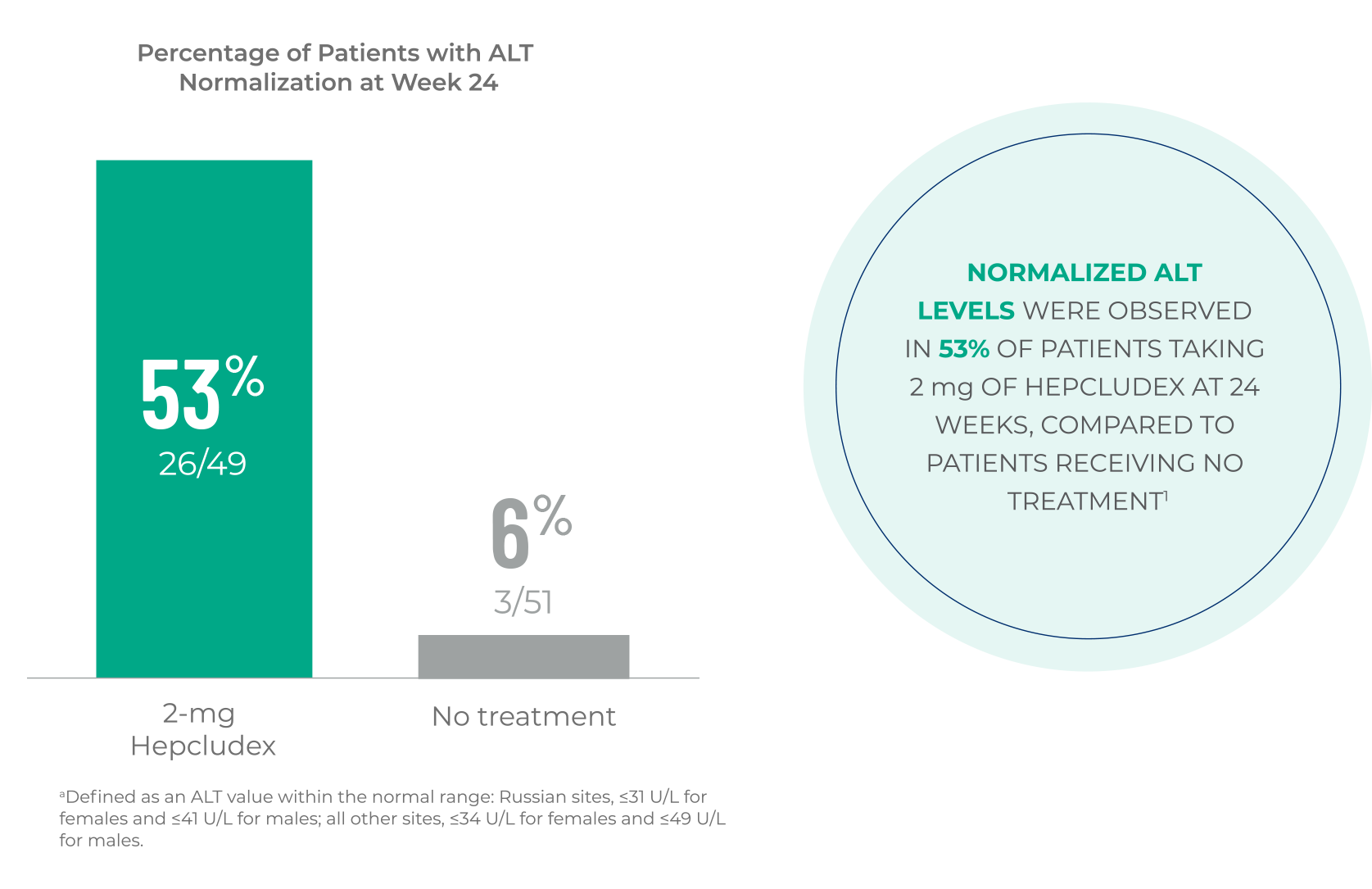
MYR301 ALT
Hepcludex demonstrated an improvement in normalized ALT at
Week 24 compared to no treatment1,a
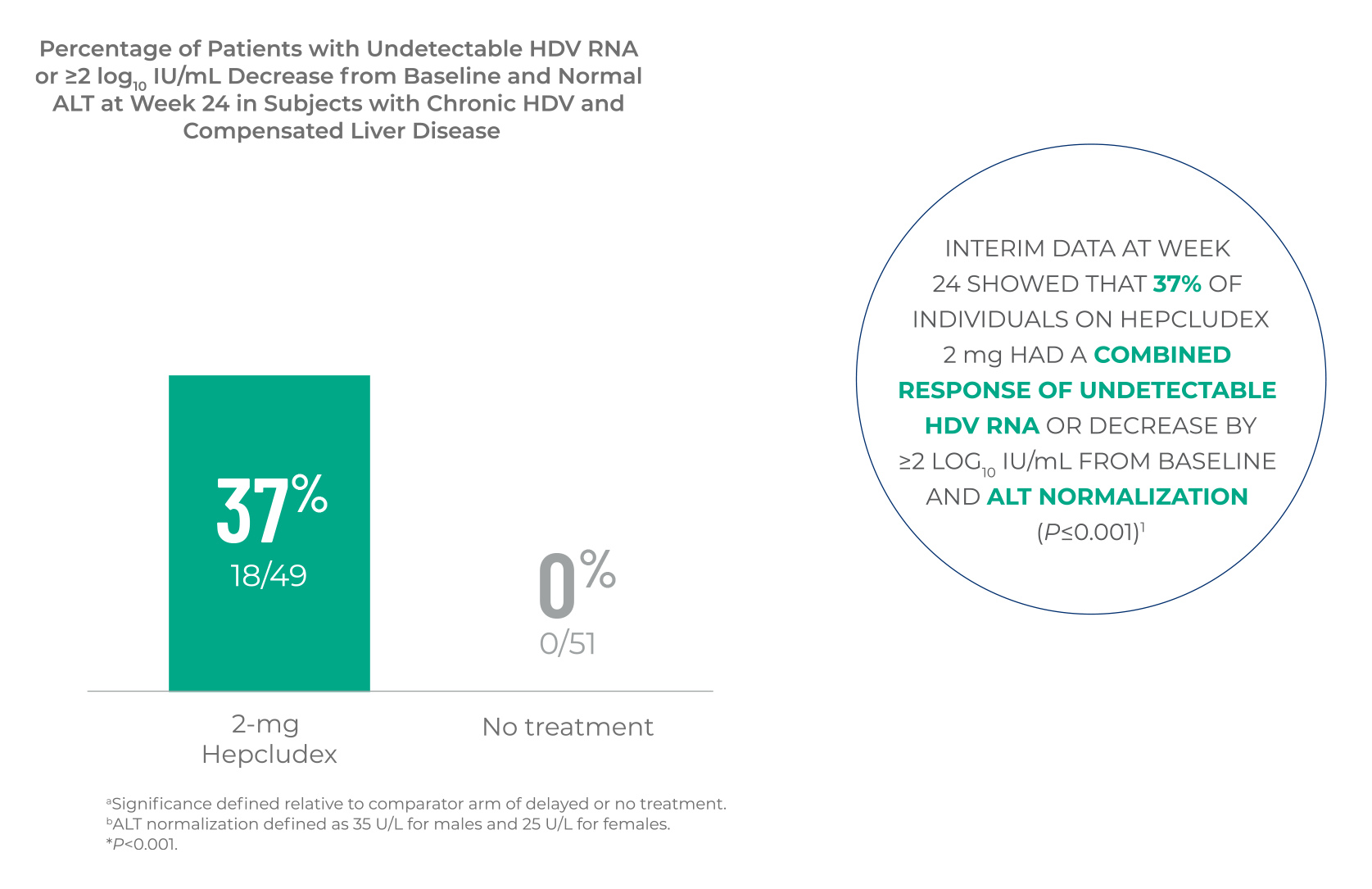
MYR301 COMBINED RESPONSE
Hepcludex demonstrated ALT normalization and a significant reduction in
HDV RNA levels at Week 24 compared to no treatment1,a,b
OVERVIEW OF ADVERSE EVENTS
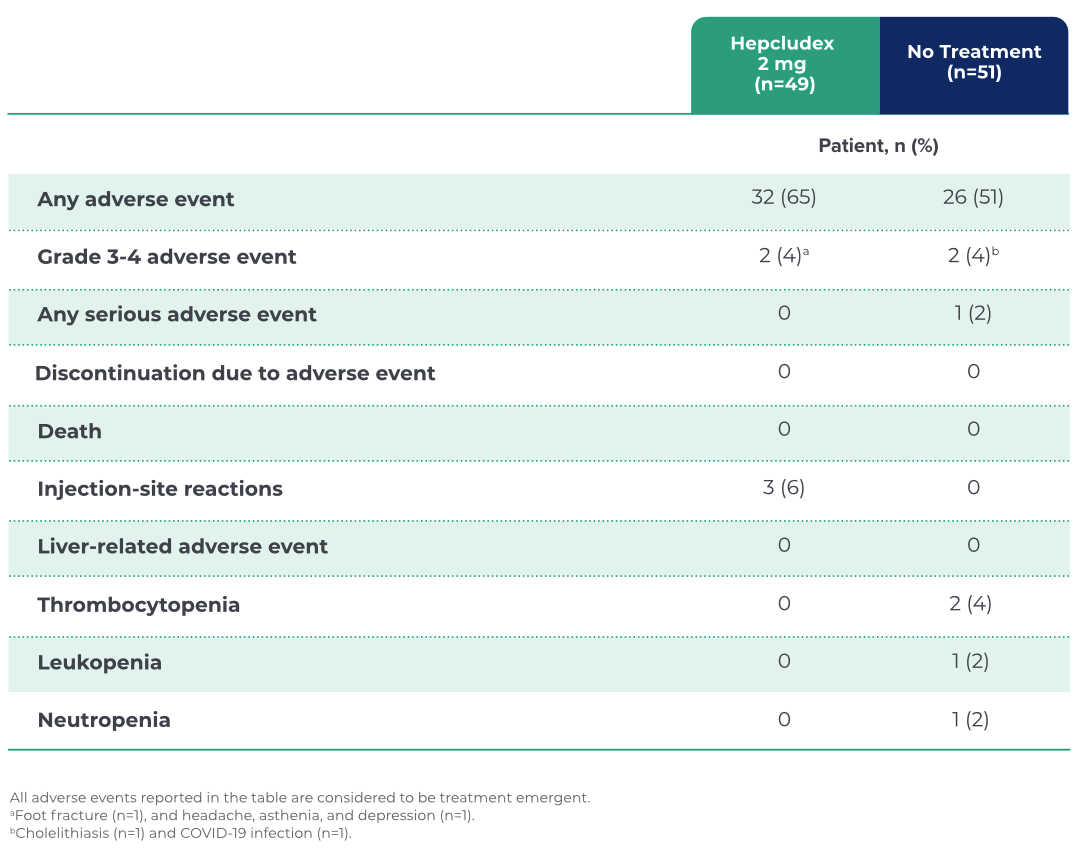
SAFETY PROFILE
Hepcludex 2 mg demonstrated a favorable safety profile with low discontinuation rates1
Injection-site reactions were
rare and mostly mild in grade11,a

No symptomatic elevations in total
bile salts were observed in the
Hepcludex 2 mg group11
REFERENCES:
1. Assessment report: Hepcludex: International non-proprietary name: bulevirtide. Procedure No. EMEA/H/C/004854/0000. May 28, 2020. Accessed November 3, 2021. https://www.ema.europa.eu/en/documents/assessment-report/hepcludex-eparpublic-assessment-report_en.pdf. 2. Wedemeyer H, Aleman S, Andreone P, et al. Bulevirtide monotherapy at low and high doses in patients with chronic hepatitis delta: 24-Week interim data of the Phase 3 MYR301 study. Poster Presentation: International Liver Congress. June 23-26, 2021. Poster # 2730.
INDICATION
Hepcludex is geïndiceerd voor de behandeling van chronische infectie met het hepatitis delta-virus (HDV) bij plasma- (of serum-) HDV-RNA-positieve volwassen patiënten met gecompenseerde leverziekte.
![]() Dit product is onderworpen aan aanvullende monitoring.
Dit product is onderworpen aan aanvullende monitoring.

Gilead Sciences Netherlands B.V.
Claude Debussylaan 22
1082 MD Amsterdam
© 2024 Gilead. All rights reserved.
HEPCLUDEX, the HEPCLUDEX logo, GILEAD, and the GILEAD logo are trademarks of Gilead Sciences, Inc.
NL-UNB-06XX
Date of production: MAY 2024

Gilead Sciences Netherlands B.V.
Claude Debussylaan 22
1082 MD Amsterdam
© 2024 Gilead. All rights reserved.
HEPCLUDEX, the HEPCLUDEX logo, GILEAD, and the GILEAD logo are trademarks of Gilead Sciences, Inc.
NL-UNB-06XX
Date of production: MAY 2024
