MYR202 STUDY INFORMATION
STUDY DESIGN
The MYR202 study was a 24-week, multicenter, open-label, randomized, Phase 2 study
in adults with chronic HDV with or without cirrhosis (n=118)1
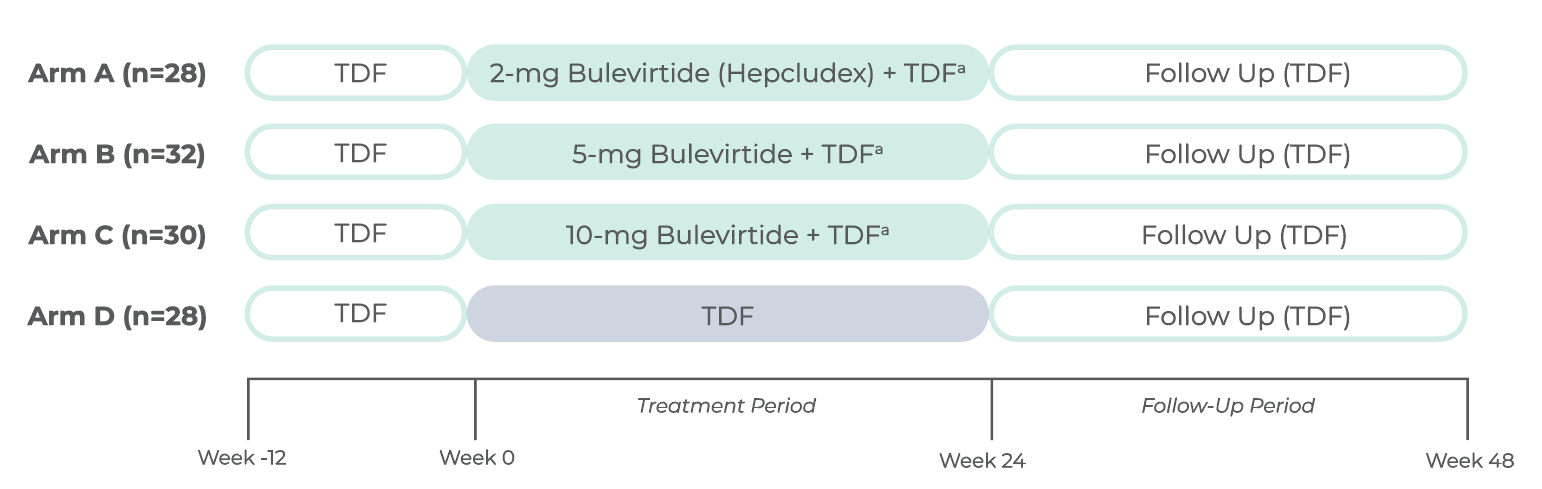
a2-mg Hepcludex is the approved dosage. Other doses have been studied as part of the dose-finding study.
TDF=tenofovir disoproxil fumarate.
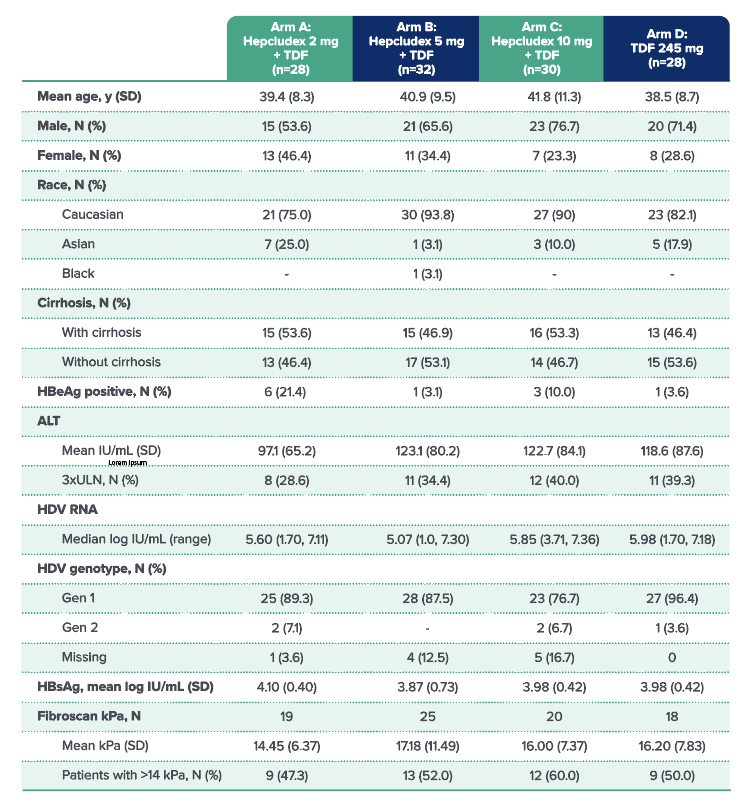
BASELINE CHARACTERISTICS

PRIMARY ENDPOINT
HDV RNA negative or a decrease in
HDV RNA by ≥2 log10 IU/mL from
baseline to Week 24
SELECT SECONDARY ENDPOINTS
- Durability of HDV RNA response to 24 weeks
post-treatment (from Week 24 to Week 48) - Combined treatment response, defined as HDV
RNA response (HDV RNA negativation or ≥2 log10
IU/mL decline) and normal ALT at treatment
Week 24 and Week 48 - Changes in ALT values at Week 24 and Week 48,
compared to baseline
PRIMARY ENDPOINT
HDV RNA negative or a decrease in
HDV RNA by ≥2 log10 IU/mL from
baseline to Week24
SELECT SECONDARY ENDPOINTS
- Durability of HDV RNA response to 24 weeks
post-treatment (fromWeek 24 toWeek 48) - Combined treatment response, defined as HDV
RNA response (HDV RNA negativation or ≥2 log10
IU/mL decline) and normal ALT at treatment
Week 24 and Week 48 - Changes in ALT values atWeek 24 and Week 48,
compared to baseline
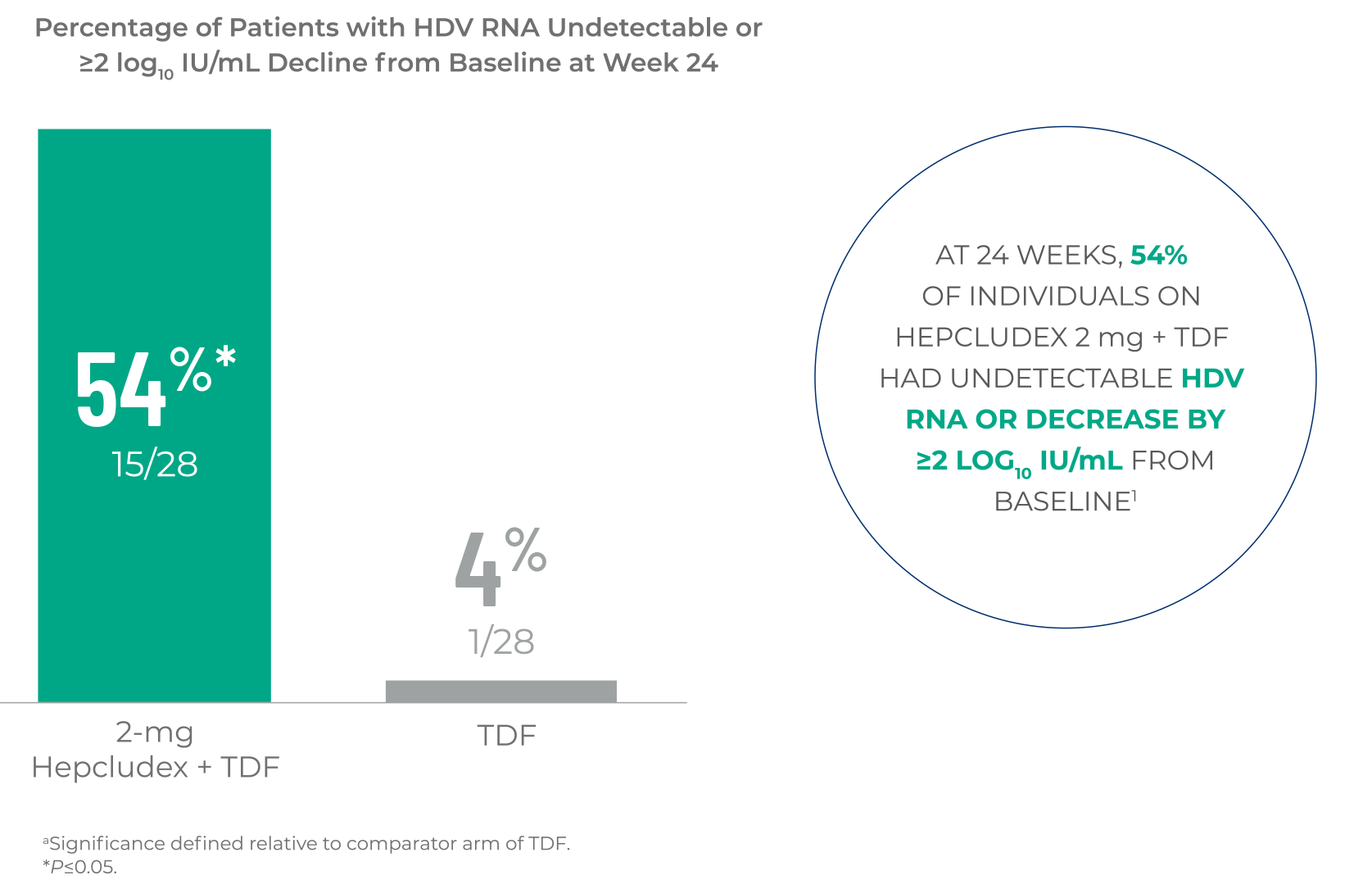
HDV RNA REDUCTION
Hepcludex demonstrated a significant reduction in HDV RNA levels at Week 24
when comparing Hepcludex + TDF to TDF alone1,a
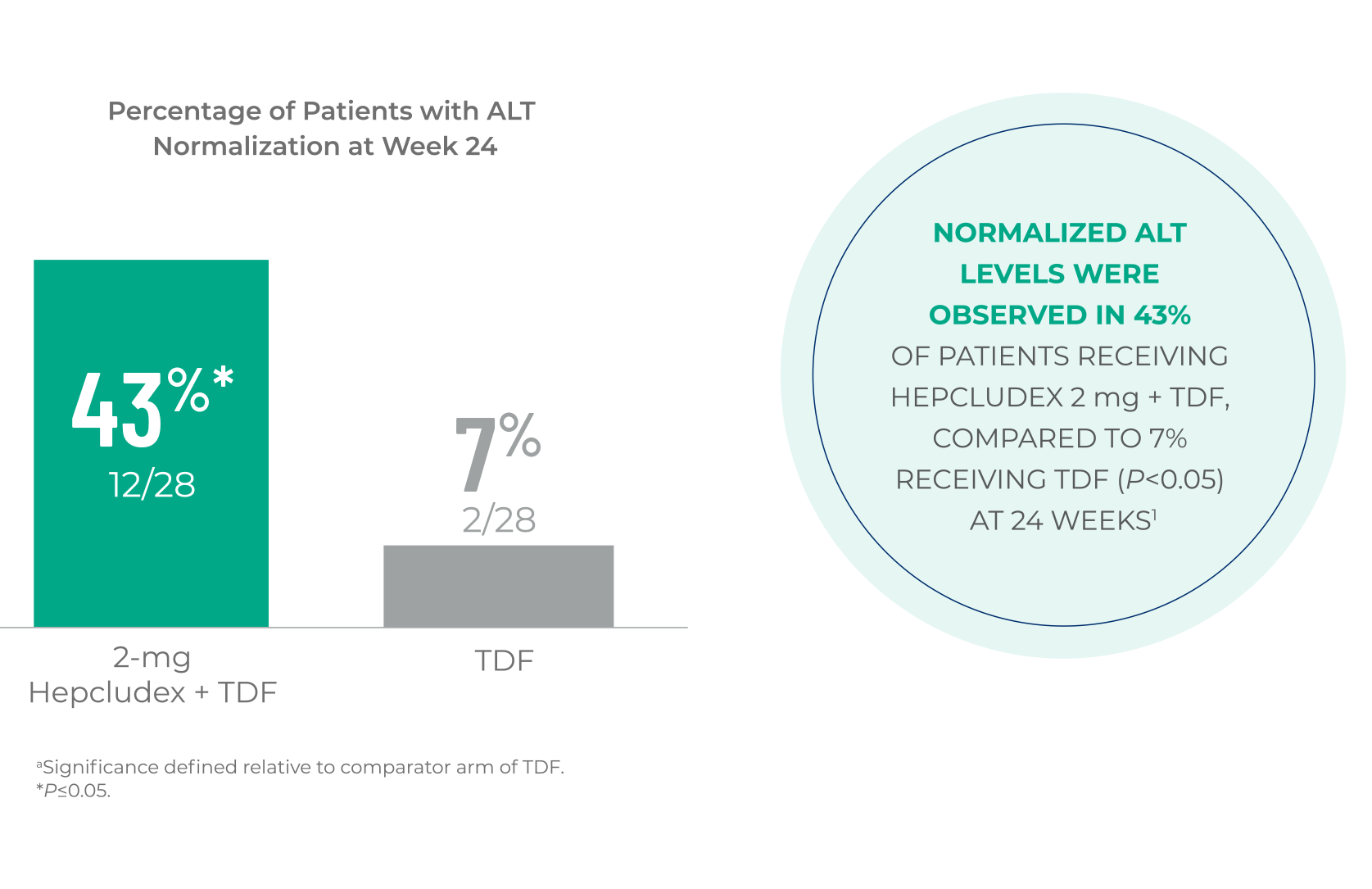
ALT NORMALIZATION
Hepcludex normalized ALT at Week 241,a
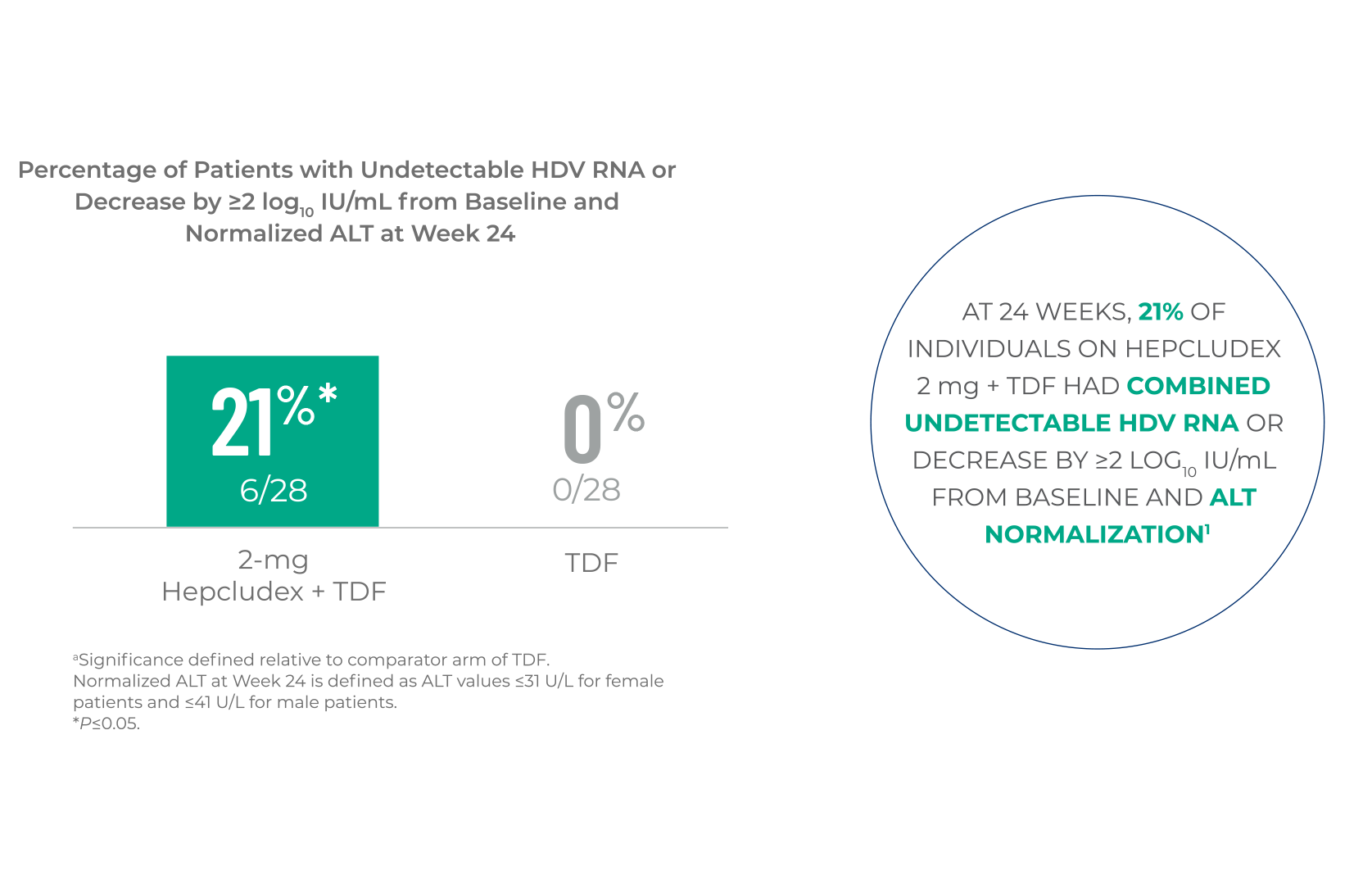
COMBINED RESPONSE
Hepcludex demonstrated a significant reduction in HDV RNA levels and improvement
in ALT normalization, compared to TDF alone at Week 241,a
OVERVIEW OF ADVERSE EVENTS
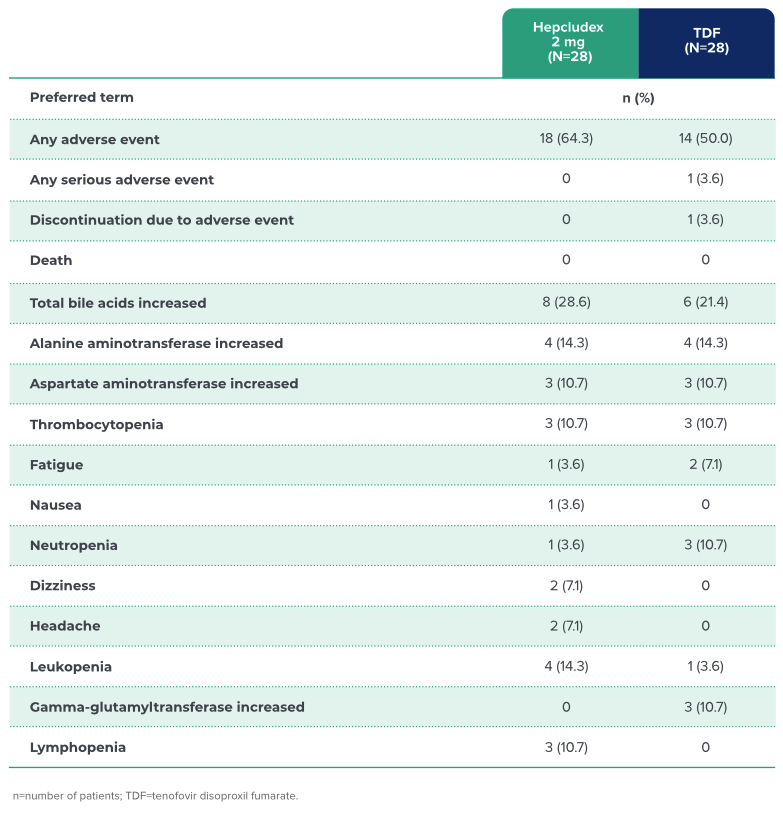
SAFETY PROFILE
Hepcludex has a demonstrated safety profile with low discontinuation rates1
In the MYR202 study, there were
no instances of treatment discontinuations
due to Hepcludex-related adverse events1,a

The most frequently reported adverse reaction
to Hepcludex was a dose-dependent,
asymptomatic increase in total bile acids and
ALT/AST elevation in the follow-up phases1
AST=aspartate aminotransferase.
a There were two study discontinuations due to AEs. All events were assessed as unrelated to the study treatment.
REFERENCES:
1. Assessment report: Hepcludex: International non-proprietary name: bulevirtide. Procedure No. EMEA/H/C/004854/0000. May 28, 2020. Accessed November 3, 2021. https://www.ema.europa.eu/en/documents/assessment-report/hepcludex-eparpublic-assessment-report_en.pdf
INDICATION
Hepcludex is geïndiceerd voor de behandeling van chronische infectie met het hepatitis delta-virus (HDV) bij plasma- (of serum-) HDV-RNA-positieve volwassen patiënten met gecompenseerde leverziekte.
![]() Dit product is onderworpen aan aanvullende monitoring.
Dit product is onderworpen aan aanvullende monitoring.

Gilead Sciences Netherlands B.V.
Claude Debussylaan 22
1082 MD Amsterdam
© 2024 Gilead. All rights reserved.
HEPCLUDEX, the HEPCLUDEX logo, GILEAD, and the GILEAD logo are trademarks of Gilead Sciences, Inc.
NL-UNB-06XX
Date of production: MAY 2024
